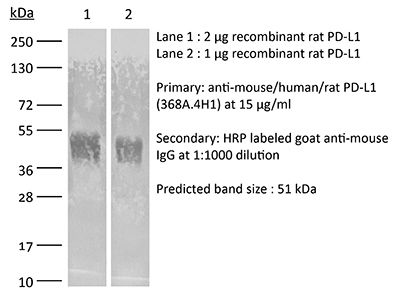
InVivoMAb anti-mouse/human/rat PD-L1
| Clone | 368A.4H1 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Catalog # | BE0383 | ||||||||||||
| Category | InVivoMab Antibodies | ||||||||||||
| Price |
|
The 368A.4H1 monoclonal antibody reacts with mouse, human, and rat PD-L1 (programmed death ligand 1) also known as B7-H1 or CD274. PD-L1 is a 40 kDa type I transmembrane protein that belongs to the B7 family of the Ig superfamily. PD-L1 is expressed on T lymphocytes, B lymphocytes, NK cells, dendritic cells, as well as IFNγ stimulated monocytes, epithelial cells and endothelial cells. PD-L1 binds to its receptor, PD-1, found on CD4 and CD8 thymocytes as well as activated T and B lymphocytes and myeloid cells. Engagement of PD-L1 with PD-1 leads to inhibition of TCR-mediated T cell proliferation and cytokine production. PD-L1 is thought to play an important role in tumor immune evasion. Induced PD-L1 expression is common in many tumors and results in increased resistance of tumor cells to CD8 T cell mediated lysis. In mouse models of melanoma, tumor growth can be transiently arrested via treatment with antibodies which block the interaction between PD-L1 and PD-1. The 368A.4H1 antibody has been shown to block the interaction between PD-L1 and PD-1.
| Isotype | Mouse IgG1, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb mouse IgG1 isotype control, unknown specificity |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Immunogen | Human PD-L1-Fc fusion protein |
| Reported Applications | in vivo PD-L1 blockade in vitro PD-L1 blockade Flow cytometry |
| Formulation | PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin | <2EU/mg (<0.002EU/μg) Determined by LAL gel clotting assay |
| Purity | >95% Determined by SDS-PAGE |
| Sterility | 0.2 μm filtration |
| Production | Purified from tissue culture supernatant in an animal free facility |
| Purification | Protein G |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
in vivo PD-L1 blockade, in vitro PD-L1 blockade, Flow cytometry
Gil Del Alcazar, C. R., et al. (2022). "Insights into Immune Escape During Tumor Evolution and Response to Immunotherapy Using a Rat Model of Breast Cancer" Cancer Immunol Res 10(6): 680-697. PubMed
Animal models are critical for the preclinical validation of cancer immunotherapies. Unfortunately, mouse breast cancer models do not faithfully reproduce the molecular subtypes and immune environment of the human disease. In particular, there are no good murine models of estrogen receptor-positive (ER+) breast cancer, the predominant subtype in patients. Here, we show that Nitroso-N-methylurea-induced mammary tumors in outbred Sprague-Dawley rats recapitulate the heterogeneity for mutational profiles, ER expression, and immune evasive mechanisms observed in human breast cancer. We demonstrate the utility of this model for preclinical studies by dissecting mechanisms of response to immunotherapy using combination TGFBR inhibition and PD-L1 blockade. Short-term treatment of early-stage tumors induced durable responses. Gene expression profiling and spatial mapping classified tumors as inflammatory and noninflammatory, and identified IFNgamma, T-cell receptor (TCR), and B-cell receptor (BCR) signaling, CD74/MHC II, and epithelium-interacting CD8+ T cells as markers of response, whereas the complement system, M2 macrophage phenotype, and translation in mitochondria were associated with resistance. We found that the expression of CD74 correlated with leukocyte fraction and TCR diversity in human breast cancer. We identified a subset of rat ER+ tumors marked by expression of antigen-processing genes that had an active immune environment and responded to treatment. A gene signature characteristic of these tumors predicted disease-free survival in patients with ER+ Luminal A breast cancer and overall survival in patients with metastatic breast cancer receiving anti-PD-L1 therapy. We demonstrate the usefulness of this preclinical model for immunotherapy and suggest examination to expand immunotherapy to a subset of patients with ER+ disease.